Publication
Article
Oncology Live®
Novel Interleukin-Based Immunotherapy Under Study in Pancreatic Cancer
Author(s):
An immunotherapy combination that adds the interleukin-10 agonist AM0010 to FOLFOX chemotherapy will be evaluated in a phase III trial for patients with metastatic pancreatic cancer that could introduce a new modality to the treatment paradigm for the malignancy.
Jeffrey R. Infante, MD
An immunotherapy combination that adds the interleukin-10 (IL-10) agonist AM0010 to FOLFOX chemotherapy will be evaluated in a phase III trial for patients with metastatic pancreatic cancer that could introduce a new modality to the treatment paradigm for the malignancy.
AM0010, a pegylated form of IL-10, stimulates an immune response by inducing the activation and proliferation of tumor-specific CD8-positive T cells, resulting in an antitumor immune memory, researchers have reported.1 Its mechanism of action is distinct from and complementary to the PD-1/PD-L1 checkpoint blockade antibodies that are rapidly gaining adoption in a wide range of cancer types, noted Jeffrey R. Infante, MD, who has conducted significant research into AM0010.
“There are no immune therapies approved for pancreatic cancer, and the initial work with PD-1 and PD-L1 inhibitors as a monotherapy has not been as promising as it is in melanoma, lung, bladder, and kidney cancers,” said Infante, director of the Drug Development Program at Sarah Cannon Research Institute in Nashville and medical oncologist at Tennessee Oncology. “If the trial is positive, this would be the first immunotherapy option for patients with pancreatic cancer.”
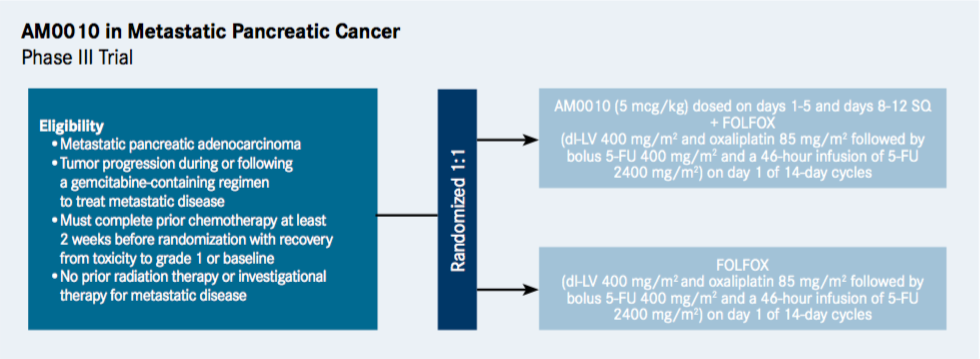
The multicenter trial, which is not yet open for enrollment, will compare the efficacy of AM0010 in combination with FOLFOX versus FOLFOX alone in an estimated 566 patients (NCT02923921).
AM0010 is administered once a day via subcutaneous injection (5 mcg/kg), while the chemotherapy is standard FOLFOX-based therapy for pancreas and colon cancer (dl-LV 400 mg/m2 and oxaliplatin 85 mg/m2 followed by bolus 5-FU 400 mg/m2 and a 46-hour infusion of 5-FU 2400 mg/m2), and is given through IV once every 2 weeks.
The trial is designed for patients 18 years and older with adenocarcinoma of the pancreas whose disease has progressed after first-line gemcitabine therapy.
The primary endpoint of the study is overall survival (OS), while the secondary endpoints are progression-free survival and overall response rate.
Earlier Clinical Trial Findings
The rationale for the trial was established in a phase I dose-escalation study for patients with advanced solid tumors including pancreatic cancer.2 Antitumor responses were assessed according to immune-related response criteria, and 41 out of the 51 enrolled patients (80.4%) were evaluable. Of the 2 evaluable patients with pancreatic cancer, 1 achieved stable disease that lasted 4 months, and both had strong reduction in the tumor marker CA19-9.2
“When we did the initial trial back in 2013, it allowed multiple tumor types, and pancreas was one of them,” said Infante, who was the principal investigator for the phase I study and is participating in the new trial. “The investigators were clearly interested in tumor sites such as renal cell, melanoma, and lung cancer that had responded to immune therapies in the past; however, they had preclinical data that suggested pancreatic cancer and a combination of AM0010 with platinum and 5-FU/leucovorin would also be an option.”
In an expansion monotherapy cohort of 22 patients with pancreatic cancer treated with AM0010 with a median of 4 prior therapies, 53% had stable disease, 27% had a more than 50% reduction of the tumor marker CA19-9, and the 1-year survival rate was 22%. Early results of a combination of AM0010 and FOLFOX showed encouraging responses; results of a phase Ib study in third-line pancreatic cancer of 21 patients will be presented at 2017 GI Cancers Symposium.
If successful when paired with FOLFOX, the combination has potential to fulfill a large, unmet need for patients with pancreatic cancer, particularly those with metastatic disease who progress from initial chemotherapy and have an OS of 6 months or less. “There’s not a large incidence rate [of pancreatic cancer] compared with other tumor types, but if you look at the people who die from their malignant disease, it’s really high,” said Infante.
The cytokine IL-10 was developed nearly 30 years ago as an anti-inflammatory agent but has presented a challenge because it has a short half-life, resulting in its being filtered quickly through the body. However, since AM0010 is a pegylated form of IL-10, it improves the exposure, and high doses of it are well tolerated.
The most common adverse events are fatigue, anemia, and thrombocytopenia, Infante said. These are usually manageable, according to Infante, particularly when looking at the “blood count issues” like anemia and thrombocytopenia.
Anemia and thrombocytopenia are also common with cytotoxic chemotherapy, and so the protocol for the phase III trial is set up to help physicians manage those toxicities, with flexibility around dose reductions and the ability to hold medications until the patient has recovered. The effect from AM0010 is rapidly reversible upon withholding the mediation, and blood counts return to normal within 2 to 3 days.
ARMO BioSciences, which is developing AM0010, is conducting the pancreatic cancer study.
References
- Infante JR, Naing A, Papadopoulos KP, et al. PEGylated IL-10 (AM0010) for advanced solid tumors—a phase 1 study. Presented at: CRI-CIMT-EATI-AACR Inaugural International Cancer Immunotherapy Conference, Translating Science Into Survival; September 16-19, 2015; New York, NY. Abstract A028.
- Naing A, Papadopoulos KP, Autio KA, et al. Safety, antitumor activity, and immune activation of pegylated recombinant human interleukin-10 (AM0010) in patients with advanced solid tumors. J Clin Oncol. 2016;34(29): 3562-3569, doi:10.1200/JCO.2016.68.1106.










